Extended lifetime for gasket materials with good chemical resistance
Lifetime is big issue for anyone selling or maintaining an equipment. At the same time, it is a rather indefinable concept. How is lifetime defined? Is it until the gasket is so worn that it can no longer be used – not really as the damage has then already taken place. Is it when the function is reduced to a given level – or something completely different?

How can you address the issue?
We are often asked how long the lifetime of a rubber component is – which is certainly a very good question. The answer always starts with: That depends on…
The lifetime depends on three factors:
- The design of the rubber component in relation to function
- The composition of the temperature and chemical conditions
- The resistivity of the rubber compound
While the first two factors are beyond our control, we have profound knowledge concerning the last factor. Resistivity may be measured against different products and chemicals at various temperatures, exposure times, etc. For a moment, let us put aside temperature dependence and focus on chemical resistance.
It goes without saying; if a rubber gasket is in contact with a medium that causes decomposition of the gasket, the lifetime will be reduced. However, it is all a little more complex when several media are involved. In order to be able to give you the best answer, most often we will carry out tests in the relevant products. An example could be food or cleaning chemicals that are often the main obstacles in the food industry. There are also a number of chemicals that represent a larger group of substances; simulants. Below is an example of four compounds used in the food industry. These compounds have been tested in chemicals that are typically used for cleaning the process equipment. As the cleaning takes place without dismantling the equipment, this process is called Clean in Place, or CIP.
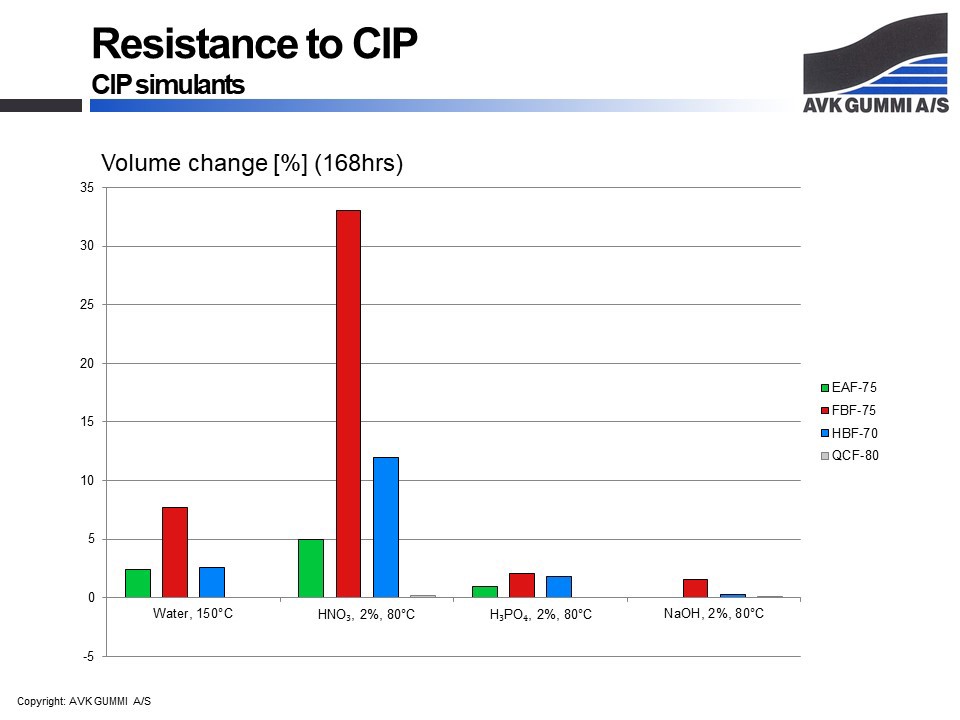
It is possible to measure various parameters; change of strength, hardness, mass and volume. Volume is one of the most decisive factors, as the gasket must fit into the component in which it is mounted.
As you can see, the volume of EAF-75 – an EPDM compound – is not changed in sodium hydroxide (NaOH), while the volume of FBF-75 – an FKM compound – is changed drastically when in contact with nitric acid (HNO3).
Typically, a cleaning process will consist of a combination of acid, base and vapour, which means that it is necessary to consider at least three different chemicals. Therefore, you will soon end up having to weigh pros and cons – a job for experts with experience in interpreting these results.
How about the temperature?
Temperature is also a decisive factor for the lifetime. It is not only a question of the normal working temperature, but just as much the maximum or minimum temperature that the gasket will be exposed to. As a rule of thumb, for each time the working temperature is increased by 10%, the lifetime will be reduced by half. As with all such principles, they are limited and should only be seen as a guideline within a relatively small temperature area.
The lifetime will also be reduced if the temperature is adequately low. The reason for this is that the material becomes more rigid as it approaches the temperature where it becomes glass-like – the glass transition temperature.

